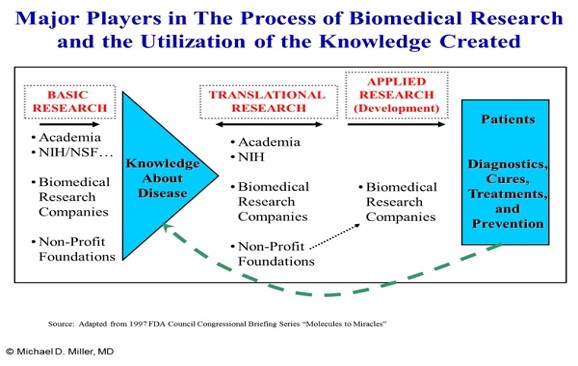
The cost of so-called specialty drugs has become a major health policy issue largely because of spending projections for new medicines for chronic hepatitis C infection and cancers. Having worked on issues related to the development, approval, availability, use, and cost of medical treatments for more than 25 years, I’ve noted with concern and bemusement how cost and value issues are being discussed and presented in public debates and policy circles.
The Intense Debate About New Biopharmaceuticals Was Predictable
The introduction of significant new specialty medicines is reasonably predictable because information about biomedical research and the developmental status of new drugs and biologics is publically available from the FDA and company press releases.…